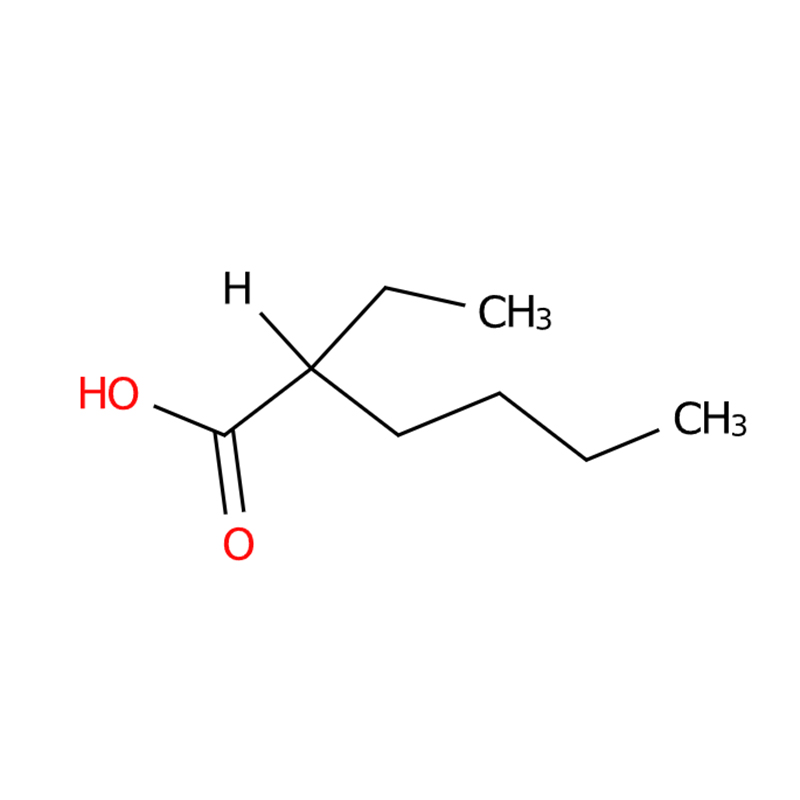
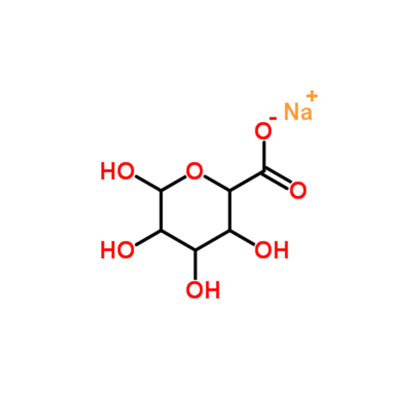
2-Ethylhexanoic acid CAS#149-57-5
Wide Industrial Versatility: 2-Ethylhexanoic acid is a key fine chemical raw material with broad applications in coatings, plastics, pharmaceuticals, pesticides, inks, and synthetic resins.
Superior Coating Performance: Its metal salts (cobalt, manganese, lead, and zinc) act as highly effective drying agents, reducing coating viscosity and producing colorless, transparent, and non-yellowing finishes—ideal for light-colored paints and varnishes.
Important Pharmaceutical Intermediate: Sodium 2-ethylhexanoate is an essential raw material for the synthesis of antibiotics such as ampicillin, while its aluminum salt is used as a surgical refrigerant and antibiotic processing aid.
Functional Additive Capabilities: It serves as a plasticizer in plastics and synthetic resins, a solvent and ester precursor in pesticides, and a thickening agent in the ink industry, enhancing processing and product performance.
2-Ethylhexanoic acid, also known as isooctanoic acid, is a carboxylic acid organic compound with the molecular formula C8H16O2 and a molecular weight of approximately 144.22. It is usually a colorless, transparent, oily liquid with a slight odor; melting point -83℃, boiling point 227.6℃, relative density 0.9105 (20℃), refractive index 1.4241 (20℃), flash point 127℃ (open cup), autoignition point 377℃, and viscosity 7.73 mPa·s (20℃); it is soluble in hot water and ether, and slightly soluble in ethanol. 2-Ethylhexanoic acid exhibits significant acidity, reacting with sodium hydroxide to form carboxylate salts; reacting with alcohols under strong oxidizing agents to form esters, such as triethylene glycol di(2-ethylhexanoic acid) ester; and reacting with alkali metals or heavy metals to form metallic soaps, such as zinc soap, lead soap, cadmium soap, and barium soap.
2-Ethylhexanoic acid Chemical Properties
| Melting point | -59 °C |
| Boiling point | 228 °C(lit.) |
| Density | 0.906 |
| Vapor density | 4.98 (vs air) |
| Vapor pressure | <0.01 mm Hg ( 20 °C) |
| Refractive index | n20/D 1.425(lit.) |
| Fp | 230 °F |
| Storage temp. | Store below +30°C. |
| Solubility | 1.4g/l |
| Form | Liquid |
| Pka | pK1:4.895 (25°C) |
| Color | Clear |
| PH | 3 (1.4g/l, H2O, 20℃) |
| Odor | Mild odour |
| PH Range | 3 at 1.4 g/l at 20 °C |
| Explosive limit | 1.04%, 135°F |
| Water Solubility | 2 g/L (20 ºC) |
| BRN | 1750468 |
| Exposure limits | ACGIH: TWA 5 mg/m3 |
| Stability | Stable. Combustible. Incompatible with strong oxidizing agents, reducing agents, bases. |
| InChIKey | OBETXYAYXDNJHR-UHFFFAOYSA-N |
| LogP | 2.7 at 25℃ |
| CAS DataBase Reference | 149-57-5(CAS DataBase Reference) |
| NIST Chemistry Reference | Hexanoic acid, 2-ethyl-(149-57-5) |
| EPA Substance Registry System | 2-Ethylhexanoic acid (149-57-5) |
| Hazard Codes | Xn |
| Risk Statements | 63-41-37/38-20/21/22 |
| Safety Statements | 36/37-36/37/39-26-2 |
| RIDADR | UN 3265 8/PG 2 |
| WGK Germany | 1 |
| RTECS | MO7700000 |
| F | 13 |
| Autoignition Temperature | 699 °F |
| TSCA | Yes |
| HazardClass | 8 |
| PackingGroup | III |
| HS Code | 29159080 |
| Hazardous Substances Data | 149-57-5(Hazardous Substances Data) |
| Toxicity | LD50 orally in Rabbit: 3640 mg/kg LD50 dermal Rabbit > 2000 mg/kg |
2-Ethylhexanoic acid is an important fine chemical raw material with wide applications in coatings, plastics, pharmaceuticals, and pesticides. In the coatings industry, cobalt, manganese, lead, and zinc salts of 2-ethylhexanoic acid are used as drying agents to reduce the viscosity of coatings, resulting in colorless, transparent, and non-yellowing coatings, making them particularly suitable for light-colored paints and varnishes. In the pharmaceutical industry, sodium 2-ethylhexanoate is a major raw material for the synthesis of the antibiotic ampicillin, and its aluminum salt can be used as a refrigerant in surgical procedures and as an adjuvant in the manufacture of antibiotics. In pesticides, 2-ethylhexanoic acid is used to prepare various ester-based fungicides and as a solvent. Furthermore, it can be used as a plasticizer in the plastics and synthetic resin industries, and as a thickener in the ink industry.