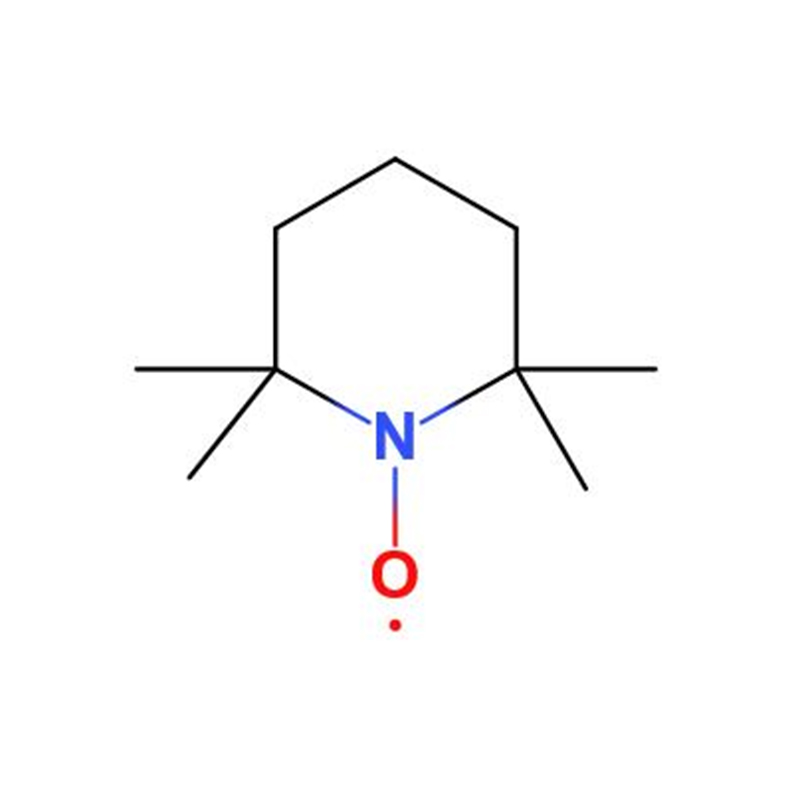
2,2,6,6-Tetramethylpiperidinooxy CAS#2564-83-2
Highly Efficient Oxidation Catalyst – TEMPO is a powerful oxidation catalyst capable of selectively converting primary alcohols into aldehydes and secondary alcohols into ketones with excellent efficiency.
Strong Free Radical Scavenging Ability – As a piperidine nitroxide free radical, it effectively captures free radicals and helps improve reaction control and stability.
Singlet Oxygen Quenching Performance – TEMPO can efficiently quench singlet oxygen, making it highly valuable in oxidation control and advanced chemical synthesis applications.
Excellent Solubility and Easy Handling – It is easily soluble in water, ethanol, benzene, and other common solvents, while its sublimable crystal or liquid form supports convenient industrial processing.
Products Description of 2,2,6,6-Tetramethylpiperidinooxy CAS#2564-83-2
2,2,6,6-Tetramethylpiperidinyl oxide, commonly abbreviated as TEMPO, is a piperidine-based nitroxide free radical. It appears as an orange-red crystal or liquid that is easily sublimable, and it dissolves readily in solvents such as water, ethanol, and benzene.
TEMPO is highly effective in capturing free radicals and quenching singlet oxygen. In addition, it serves as an efficient oxidation catalyst, selectively oxidizing primary alcohols to aldehydes and secondary alcohols to ketones.
2,2,6,6-Tetramethylpiperidinooxy Chemical Properties
| Melting point | 36-38 °C(lit.) |
| Boiling point | 193°C |
| Density | 1 g/cm3 |
| Vapor pressure | 0.4 hPa (20 °C) |
| Refractive index | 1.4350 (estimate) |
| Fp | 154 °F |
| Storage temp | Store below +30°C. |
| Solubility | 9.7g/l |
| Form | Powder |
| Color | Yellow to green |
| PH | 8.3 (9g/l, H2O, 20℃) |
| Water Solubility | Soluble in all organic solvents. Insoluble in water. |
| Merck | 149140 |
| BRN | 1422418 |
| Stability: | Stable. Incompatible with strong acids, strong oxidizing agents. Refrigerate. |
| InChIKey | RVWUHFFPEOKYLB-UHFFFAOYSA-N |
| CAS DataBase Reference | 2564-83-2(CAS DataBase Reference) |
| NIST Chemistry Reference | 1-Piperidinyloxy, 2,2,6,6-tetramethyl-(2564-83-2) |
| EPA Substance Registry System | 1-Piperidinyloxy, 2,2,6,6-tetramethyl- (2564-83-2) |
Safety Information
| Hazard Codes | C,Xi |
| Risk Statements | 34-36/37/38 |
| Safety Statements | 26-36/37/39-45-24/25 |
| RIDADR | UN 3263 8/PG 2 |
| WGK Germany | 3 |
| RTECS | TN8991900 |
| Autoignition Temperature | 275 °C |
| TSCA | Yes |
| HazardClass | 8 |
| PackingGroup | III |
| HS Code | 29333999 |
Product Application of 2,2,6,6-Tetramethylpiperidinooxy CAS#2564-83-2
TEMPO is widely used in chemistry, biology, the food industry, agriculture, and many other fields. It functions as a free radical scavenger, singlet oxygen quencher, and selective oxidation catalyst.
In polymer chemistry, it is applied as a free radical scavenger, inhibitor, anti-aging agent, thermal degradation inhibitor, as well as a light and heat stabilizer. It can also react with active chain free radicals to form covalent dormant species, which can later split back into chain free radicals and continue chain growth.
In organic synthesis, TEMPO is commonly used as a catalyst for the oxidation of various alcohols and polyols. It selectively oxidizes primary alcohols to aldehydes without further oxidation to carboxylic acids, while secondary alcohols are oxidized to ketones. The TEMPO/NaClO/NaBr system is a recyclable oxidation system in which the actual oxidizing species is the N-oxoammonium ion formed by oxidation of TEMPO with NaClO. In this cycle, the hydroxyl group is oxidized while TEMPO is regenerated by the co-oxidant, allowing the oxidation process to continue efficiently. In addition, many other TEMPO-based oxidation systems are widely used in synthesis.